A large number of cultural properties were damaged in the March 2011 tsunami that devastated Japan’s Tohoku region. A problem arises when we are faced with removing salt matter from paintings: salts absorb moisture from the atmosphere, and that moisture can promote deformation of the support. The Tokyo National Museum began in 2011 to research methods for removal of salt matter from paintings on canvas. Through a number of experiments, methods of desalination were developed for acrylic paintings on cotton canvas. In one method, the canvas was temporarily removed from its original wooden frame, edge-lined with polyester cloth, and stretched on a temporary frame. Water was sprayed on the reverse and moisture absorbed with blotting paper. Experiments that followed confirmed that the amount of moisture and the period of immersion influenced the removal of remaining salts. Using techniques from paper conservation, moistened blotting paper was used as a compress, and water containing dissolved salts was removed as quickly as possible using polymer sheets. This process was repeated several times for desalination. As a result, it was possible to control the contraction of the canvas to about 0.3%, and the chloride concentration was reduced to about what is contained in tap water in Tokyo.
63. Stabilization Processing of Canvas Paintings Damaged by Tsunami: Consideration of Blotter Washing to Acrylic Paintings on Cotton Canvas
- Yuko Tsuchiya, Painting Conservator, Tokyo University of the Arts
- Eriko Hoshi, Conservation Scientist, Joshibi University of Art and Design, Tokyo
- Hiroshi Haze, Painting Conservator, Haze Painting Studio, Tokyo
- Marie Moto, Paper Conservator, Japan Conservation Project
- Nobuyuki Kamba, Conservation Scientist, Tokyo National Museum
Background
A large number of cultural properties were damaged in the March 2011 tsunami, an unprecedented natural disaster that devastated Japan’s Tohoku region (fig. 63.1). The process of conserving these properties has lasted nine years and remains ongoing to this day. At that time, I (Tsuchiya) was working at the Conservation and Preservation Department of Tokyo National Museum, and I was put in charge of looking into how to desalinate canvas paintings as part of the second stage of the rescue process. Specifically, I was entrusted with the task of restoring fifty-three canvas paintings from Rikuzentakata City Museum. These paintings were wrapped in plastic coverings at the time of the disaster (fig. 63.2). After the tsunami, they absorbed large volumes of grime and moisture and were kept in this moist state. As a result, mold grew across the surfaces, and there was a considerable amount of paint flaking.


As a first step in the rescue process, remedial treatment was undertaken to remove the surface grime and mold and to fumigate the paintings (Ito, Yumi. 2014. “Immediate Treatment of Oil Paintings.” In Stabilization Processing: Multi-Organizational Co-Operative Project for Preserving and Restoring Cultural Assets Damaged by Tsunami on March 11th, 2011, edited by Lois Lydens, 106–9. Tokyo: ICOM Japan.). However, it was impossible to remove the embedded grime and salt without using water, so those were left untreated during this stage. Chloride compounds left on the canvases after the tsunami ionized, and a certain amount of moisture was needed to remove them. Given the assumption that canvas shrinkage is a major factor impacting the status of the paints, the question arose as to how much shrinkage was tolerable. Based on research already carried out overseas, the shrinkage rate was set at within 0.5% (Mecklenburg, Marion F. 2007a. “Determining the Acceptable Ranges of Relative Humidity and Temperature in Museums and Galleries, Part 1: Structural Response to Relative Humidity.” Report, Smithsonian Museum Conservation Institute, Washington, DC. https://repository.si.edu/handle/10088/7056., Mecklenburg, Marion F. 2007b. “Determining the Acceptable Ranges of Relative Humidity and Temperature in Museums and Galleries: Part 2, Structural Response to Temperature.” Report, Smithsonian Museum Conservation Institute, Washington, DC. https://repository.si.edu/handle/10088/7055.), and the research team set a goal of minimizing the damage to the paint layer by minimizing the shrinkage as much as possible.
After carrying out several experiments, our research focused on acrylic paintings painted on cotton canvases. We made several attempts to clean these paintings using water. In the end, we decided to adopt a desalination method that used the blotting technique when removing salt from the acrylic paintings (Tsuchiya, Yuko, Eriko Hoshi, Marie Moto, Hiroshi Haze, and Nobuyuki Kamba. 2017. “Stabilization of Canvas Paintings Damaged by Tsunami: Desalination of Blotter Washing to Acrylic Paintings on Cotton Canvas.” In Japan Society for the Conservation of Cultural Property 39th Conference Preprints, Kanazawa (Japan), 1–2 July 2017, 28–29.). We essentially achieved our target with regard to the amount of chlorine remaining after treatment (Tsuchiya, Yuko. 2018. “Stabilization Treatment for Acrylic Paintings on Cotton Cloth.” Stabilization Processing: Multi-organizational Co-operative Project for Preserving and Restoring Cultural Assets Damaged by Tsunami on March 11th, 2011, 33–42. Enlarged edition. Tokyo: ICOM Japan.).
Desalination Process
The desalination work to remove the embedded grime and salt from the canvases could not be undertaken with any solvent besides water. However, there was a danger that water might cause the canvas to shrink or the surface paints to peel off. We focused on the conservation state of fifteen large acrylic paintings among those canvas paintings damaged by the tsunami.
Hardly any of the wooden frames were warped, even with the larger paintings, while the canvases had only minimal shrinkage and there was no paint flaking. As a result, it was reasoned that shrinkage could probably be kept to a minimum even if water was applied to these canvases. We prepared a life-size mock-up of a large acrylic painting, stretched it over an actual stretcher, and applied water. Hardly any shrinkage was observed (Tsuchiya, Yuko, Nobuyuki Kamba, Hiroshi Wada, Tominori Araki, Haruhiko Suzuki, Otoyo Yonekura, Tomoe Hirakawa, Ayako Ogawa, Hiroshi Haze, Norie Nishihara, and Kumi Ikegam. 2014. “Conservation Treatment for Recovery Efforts after the Disaster with Cultural Assets: Experiments of Desalination for Canvas Paintings.” In Japan Society for the Conservation of Cultural Property 36th Conference Preprints, Tokyo (Japan), 7–8 June 2014, 182–83.).
Based on this, we undertook a desalination trial using the absolute minimum amount of moisture. We first tried a treatment method using gel sheets. Gel sheets with a 1.5% concentration and a 7:2:1 ratio of carrageenan (a seaweed-based emulsifying agent), xanthan gum, and locust bean gum were chosen for their ease of use and suitability for the syneresis process. A 16 × 11 × 2 cm sheet was pressed against the rear side of the canvas and left for about thirty minutes before being replaced with a new sheet. This process was carried out twice. However, the chlorine (CI) values of the canvas (rear side) remained at 50%–80% of the initial values (Tsuchiya, Yuko, Hiroshi Haze, Eriko Nakau, Kumi Masuda, Marie Moto, Hiroshi Wada, Tominori Araki, and Nobuyuki Kamba. 2015. “Conservation Treatment for Recovery Efforts after the Disaster with Cultural Assets: Experiments of Desalination for Paintings on Linen Canvas.” In Japan Society for the Conservation of Cultural Property 37th Conference Preprints, Kyoto (Japan), 27–28 June 2015, 86–87.). This was because the gel sheets alone did not have sufficient moisture to remove the salt and other material that had seeped into the canvas fiber.
We next examined the blotter-washing technique used in paper conservation. After several experiments, we reached the conclusion that this method could be used to desalinate the acrylic paintings among the canvas paintings in Rikuzentakata City Museum’s collection, so we carried out the conservation work accordingly.
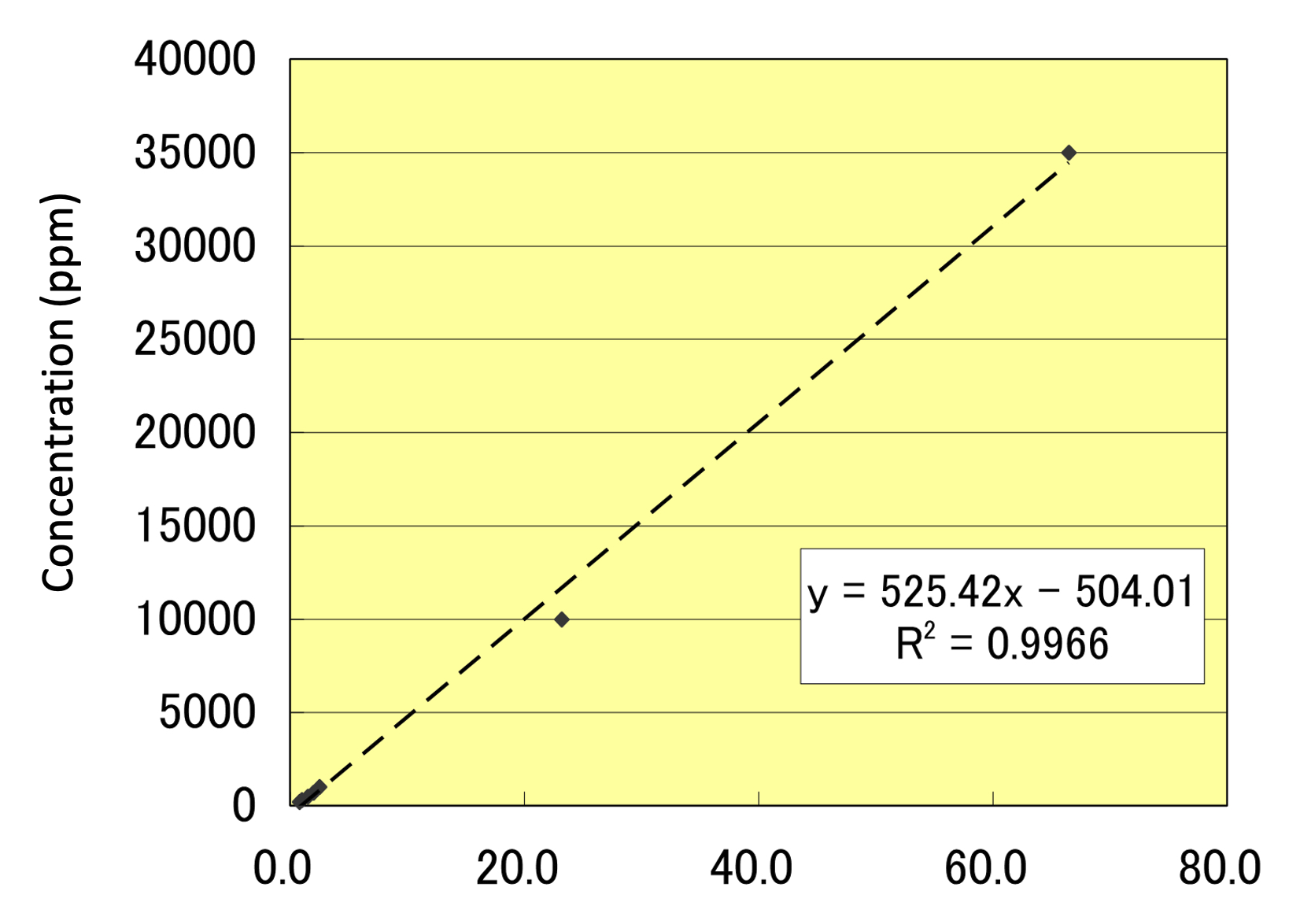
Preparing a Calibration Curve for the Cotton Cloth Used for the Canvas
The residual chlorine CI values were measured using X-ray fluorescence spectroscopy (XRF). The obtained values were in centipoise (cP), so a calibration curve was prepared to convert these into parts-per-million (ppm) values. As the affected acrylic painting canvas was on cotton, we prepared a cotton cloth for our trials. We also prepared CI water solutions of 1, 10, 50, 100, 200, 300, 500, 700, 1000, 10,000, 35,000, and 100,000 ppm. These were applied to pieces of the cotton cloth and then dried. These were set as reference standards and measured using XRF. We targeted a figure of 33 ppm (the CI value of drinking water in Tokyo) for the residual CI density on the post-treatment canvas (fig. 63.3).
The Desalination Work
The target work was an acrylic painting by Masayoshi Nameki. The canvas had already been removed from the original stretcher. Polyester strips were attached to the edges of the canvas (strip-lining), and the canvas was remounted on a larger stretcher. The size of the canvas was recorded before applying water, then the desalination process was carried out as follows:
-
Water mixed with ethyl alcohol was sprayed onto the back of the canvas to apply moisture to the entire surface (fig. 63.4).

Figure 63.4 Water mixed with ethyl alcohol was sprayed onto the back of the canvas to apply moisture to the entire surface. Image: Research team -
The entire surface was covered with dampened blotting paper and left for eight minutes (fig. 63.5).

Figure 63.5 The entire surface was covered with dampened blotting paper and left for 8 minutes. Image: Research team -
A highly absorbent resin sheet was placed over the blotting paper for two minutes to soak up the grime and salt absorbed by the blotting paper.
-
The blotting paper and resin sheet were removed and steps 1–3 were repeated five or six times. After the first application, the water that was sprayed on did not contain alcohol.
-
A thin paper sheet was placed against the surface and sprayed with water. This process was carried out twice to remove the absorbed grime and salt.
-
The salinity concentration levels of the chloride test paper were measured each time (fig. 63.6, table 63.1).
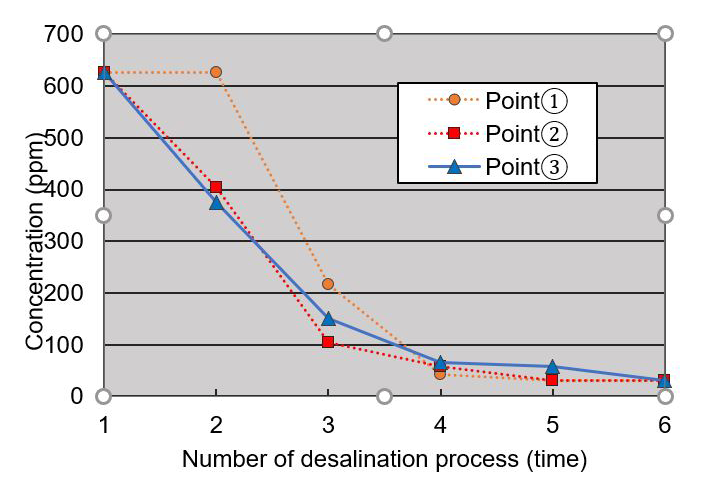
Figure 63.6 Salinity concentration levels of the chloride test paper were measured after each application of water (see table 63.1). Image: Research team -
Canvas shrinkage was recorded directly after each application of water, after the desalination process was totally finished, and the following day, when the canvas had dried.
Conclusion and Future Issues
The blotting method can be used to remove residual salt from acrylic paintings painted thinly on cotton-fiber canvases. The shrinkage rate of the canvas can be kept to within 0.5% of the total surface by maintaining the painting under tension during the treatment (tables 63.2, 63.3).
| Process | Condition | Date of work | Short-side length (weft) (mm) | Expansion and contraction (mm) | Rate of change (%)* | Long-side length (warp) (mm) | Expansion and contraction (mm) | Rate of change (%) | |
|---|---|---|---|---|---|---|---|---|---|
| Before treatments | Dry | 5/29 | 1817 | 2272 | |||||
| Mounting onto a temporary frame | After moving from wood frame | Dry | 5/29 | 1810 | −7 | 0.4 | 2263 | −9 | 0.4 |
| After temporary mounting | Dry | 5/30 | 1817 | +7 | 0.4 | 2269 | +6 | 0.3 | |
| Desalination work | Before the work | Dry | 7/12 | 1819 | +2 | 0.1 | 2273 | +4 | 0.2 |
| After the work† | Wet | 7/12 | 1798 | −19 | 1.2 | 2263 | −10 | 0.4 | |
| 1 day after | Dry | 7/13 | 1815 | +17 | 0.9 | 2270 | +7 | 0.3 | |
| 2 days after | Dry | 7/14 | 1815 | 0 | 0.0 | 2270 | 0 | 0 | |
| 2 weeks after | Dry | 7/26 | 1818 | +3 | 0.2 | 2270 | 0 | 0 |
*Rate of change in dimension before treatments.
†1 hour after drying by exchanging blotting paper three times.
| Process | Condition | Date of Work | Short-side length (weft) (mm) | Expansion and contraction (mm) | Rate of change (%)* | Long-side length (warp) (mm) | Expansion and Contraction (mm) | Rate of change (%) | |
|---|---|---|---|---|---|---|---|---|---|
| Before treatments | Dry | 5/29 | 1818 | 2272 | |||||
| Mounting onto a temporary frame | After moving from wood frame | Dry | 5/31 | 1810 | −8 | 0.4 | 2263 | −8 | 0.4 |
| After temporary mounting | Dry | 5/31 | 1817 | +7 | 0.4 | 2269 | +5 | 0.2 | |
| Desalination work | Before the work | Dry | 7/13 | 1819 | +2 | 0.1 | 2273 | +2 | 0.1 |
| During the work | Wet | 7/13 | 1803 | −16 | 0.9 | 2265 | −7 | 0.3 | |
| Immediately after the work | Wet | 7/13 | 1799 | − | 0.2 | 2265 | 0 | 0 | |
| After the work | Wet | 7/13 | 1799 | 0 | 0 | 2265 | 0 | 0 | |
| After the work | Wet | 7/13 | 1798 | −1 | 0.1 | 2264 | −1 | 0 | |
| 1 day after | Dry | 7/14 | 1819 | +21 | 1.2 | 2273 | +9 | 0.4 | |
| 2 weeks after | Dry | 7/26 | 1818 | −1 | 0.1 | 2273 | 0 | 0 |
*Rate of change in dimension before treatments.
Researchers outside of Japan have been aware of the dangers of using excess moisture when cleaning paintings since the 1980s. In particular, there has been considerable debate about problems involving acrylic paints (a relatively new painting material), such as the swelling of resins due to the impact of various additives ( Jablonski, Elizabeth, Tom Learner, James Hayes, and Mark Golden. 2003. “Conservation Concerns for Acrylic Emulsion Paints.” Reviews in Conservation 48, supp. 1: 3–12.; Tumosa, Charles S., and Marion F. Mecklenburg. 2004. “Moisture Isotherms of Acrylic Emulsion Paints.” WAAC Newsletter 26, no. 3: 12–14.; Ormsby, Bronwyn, and Tom Learner. 2009. “The Effects of Wet Surface Cleaning Treatments on Acrylic Emulsion Artists’ Paints: A Review of Recent Scientific Research.” Reviews in Conservation 54, supp1: 29–41. https://doi.org/10.1179/sic.2009.54.Supplement-1.29.; Doménech-Carbó, María Teresa, Miguel F. Silva, Elvira Aura-Castro, Antonio Doménech-Carbó, Laura Fuster-López, Jose V. Gimeno-Adelantado, Stephan U. Kröner, María Luisa Martínez-Bazán, Xavier Más-Barberá, Marion F. Mecklenburg, Laura Osete-Cortina, and Dolores J. Yusá-Marco. 2013. “Multitechnique Approach to Evaluate Cleaning Treatments for Acrylic and Polyvinyl Acetate Paints.” In New Insights into the Cleaning of Paintings: Proceedings from the Cleaning 2010 International Conference, Universidad Politecnica de Valencia and Museum Conservation Institute, edited by Marion F. Mecklenburg, A. Elena Charola, and Robert J. Koestler, 125–34. Washington, DC: Smithsonian Institution Scholarly Press.; Toriumi, Hidemi. 2018. “Development of Cleaning Method for Paintings in Europe and the United States since the 1980s.” In Japan Society for the Conservation of Cultural Property 40th Conference Preprints, Kyoto (Japan), 16–17 June 2018, 280–81.; Hackney, Stephen. 2020. On Canvas: Preserving the Structure of Paintings. Los Angeles: Getty Publications.). Additive agents in acrylic paints tend to rise to the surface of paints, so these paintings probably lost these additives as a result of the tsunami. Furthermore, a water-based treatment was also used during the desalination process. In light of these factors, going forward we will need to ascertain the damage to the paints by carrying out experiments while preparing specimens.
Acrylic paints are a new painting material, so we still have a lot to learn with regard to the deterioration process. While searching for a way to remove grime without damaging the paint layer, we need to constantly carry out post-treatment observations and the like to check for any problems with the method we adopted.
Further research will be needed to determine the extent to which this method can be applied to oil paintings on linen canvases.
Acknowledgments
This research was carried out with support from the National Task Force for the Japanese Cultural Heritage Disaster Risk Mitigation Network’s Cutting Edge Scientific Research into Disaster Risk Mitigation, a subsidy provided by the Agency for Cultural Affairs. Technological advice regarding the blotting process was provided by Norie Nishihara and Otoyo Yonekura. We would also like to thank the Smithsonian Institution’s Marion Mecklenberg for providing invaluable advice about canvas shrinkage.