Additional Contributors: Aurélia Azéma, Ann Boulton, Manon Castelle, Jens Daehner, Joachim Kreutner, Emmanuel Lamouche, Annick Texier, Brice Vincent
This chapter summarizes the different types of and , how to identify them, and what technical analysis can assist.
1 What are gilding and plating?
Different metals can be applied to the surface of bronze sculptures to produce polychromatic effects, to imitate precious metal, or as protection against . ( and are other ways of applying metal to the surface; see I.9.) Many metals in addition to gold can be used, including platinum, silver, copper, and aluminum. This is broadly called “plating,” and more specifically “gilding” when the applied metal is gold. Thus, the presence (or absence) of plating, the identification of metal used, and the method of attachment or adhesion are key elements in the technical study of any sculpture.
1.1 What are the most common gilding techniques on bronze
sculpture?
1.1.1 Leaf gilding
The application of very thin sheets, known as leaf, is a common technique requiring very little precious metal.1 Gold leaf is typically 0.1–1.2 µm thick,2 normally 7–11 cm square, although smaller pieces are encountered (fig. 287).3 Found as early as the second millennium BCE in Egypt, gold leaf was widely used in the Greek and Roman world (fig. 288), then declined in the West during the medieval period due to the development of mercury gilding, then gained popularity again in the Renaissance.4 In Asia it is still used for religious sculptures, and devotees apply gold leaf to Buddha figures as an act of worship (fig. 289).5 Leaf may be applied directly to clean metal or attached with adhesive.6
Variants
Three prevalent adhesive techniques are used for applying metal leaf to bronze sculpture: water gilding, oil gilding, and lacquer gilding. Gold leaf can also be applied using mercury, as described below (I.7§1.1.2). Little is known about the adhesives used on ancient sculptures because they degrade easily. In Asia, the use of lacquer is well known.7 In the West the choice of adhesive is more complex, with a distinction generally made between water gilding and oil gilding (terminology originating from traditional wood gilding). Water gilding normally has an animal-glue adhesive over a gesso ground that is sometimes covered with a clay layer to allow the gold to be burnished for a more compact and reflective surface. This process was used in ancient Mesopotamia and Egypt, but on bronze it is uncommon in later periods.8 Oil gilding is a process in which gold leaf is adhered to the metal surface using an oil-based adhesive; it is more suited to outdoor conditions than water gilding. Many metals in addition to gold can be applied by the oil technique, including platinum, silver, copper, and aluminum.
Identification
-
Leaf gilding is very thin and therefore delicate.
-
It is usually golden yellow because of the high gold content (about 90–98%).9
-
The prime indicator that leaf has been used is the overlapping of the straight edges, visible as squares on the surface (figs. 287, 290). With wear, the overlapping edges are sometimes all that remains because of the greater thickness of material.
-
Leaf has a tendency to flake off as the adhesive ages.
-
Traces of a gesso may indicate that gilding was once present.
-
Traditionally, the siccative (drying agent) added in the oil gilding process was a lead compound such as litharge (lead oxide). In the twentieth century, lead was gradually replaced by cobalt, manganese, and iron compounds, which can be detected by elemental analysis.
-
For the gilding and regilding of outdoor sculptures, yellow epoxy paint is often applied as a preparation layer to prevent corrosion of the bronze, but also to hide wear. Its presence is an indicator of modern gilding.
Risks of misidentification/misinterpretation
-
Dutch leaf, around 0.3 µm thick and made of , is sometimes used in imitation of gold leaf. It may be detected because it darkens (tarnishes), and elemental analysis such as X-ray fluorescence spectroscopy (XRF) can be used to further distinguish it.
-
Gold-colored paint, often used in restoration, can be identified under magnification by its granular appearance (see I.8§1.2.1). This may be made with powdered gold or brass in a binder. Modern “gold” paints may contain nonmetallic luster pigments.
1.1.2 Mercury gilding (also known as fire gilding or
amalgam gilding)
Gold (and less commonly silver or tin) can be amalgamated with mercury to form a metallic paste to apply to the casting. The gilded metal is heated to about 250–400°C to vaporize the free mercury, leaving a matte layer of gold.10 The resulting porous surface is then usually burnished to form a compact layer. If the substrate is carefully prepared prior to gilding, burnishing can produce a highly reflective and durable surface, with the gold effectively fused with the surface of the bronze. A further process known as mise en couleur was used at least as early as the Renaissance period to alter the reflective qualities and color of the gilding by applying various chemicals. Historical texts describe the process, which involved heating chemicals such as sulfur, saltpeter, and other metal salts.11
Mercury gilding has been widely used on sculpture (fig. 291), appearing in China as early as the Warring States period (475–221 BCE) (fig. 292).12 In Europe, it can be found a little later, in the Late Iron Age and Roman periods, flourishing in the Middle Ages, the Renaissance,13 and into the nineteenth century.14 Safety legislation brought its use almost completely to an end in Europe, though not in all parts of the world,15 by the close of the nineteenth century, due to the toxicity of the mercury vapors. Because of the semiliquid application, it is more suited to gilding complex shapes than foil or leaf. It has also been used to gild selected parts of an object (known as parcel gilding) (fig. 293).
Variants
An alternative method is to apply mercury directly onto the surface of the object, lay on gold leaf, and then heat.16
Identification
-
The detection of mercury is the key identifier: mercury compounds remain in the gilding even after the free mercury is driven off.17
-
Amalgam gilding is applied as a semiliquid, so may show splashes or runs (figs. 293, 294), and adherence to the bronze substrate is good, though the gilding may show wear at high points.
-
The thickness of the gilding layer can vary from 1 to 10 µm.18
-
A metallographic cross section of mercury gilding will show good bonding to the bronze substrate (fig. 295).
-
The interior of a sculpture is never fully gilded, though drips may be present.
-
If the lead content of a sculpture exceeds 1% it has been thought unlikely that the surface was mercury gilded because lead globules on the surface spoil the gilding,19 but this does not seem to be a universal rule.20
Risks of misidentification/misinterpretation
-
Mercury may be present on a sculpture when there is no mercury gilding, for example if the red pigment cinnabar (mercury sulfide) is present. This was observed on some Tibetan bronzes that were gilded using other techniques.21
-
Minute traces of mercury may be present as residues of some metal polishes,22 or from quicking for electroplating (see I.7§1.1.3 below), although these traces are usually too small to be detected by XRF.
1.1.3 Electroplating (also known as electrogilding or
voltaic gilding)
Electroplating was developed in the first half of the nineteenth century.23 It relies on an external electrical current to dissolve the plating metal (the anode) and move the ions through an electrically conductive electrolyte solution to the object to be plated (the cathode). It has good adhesion and is able to consistently produce a very thin, continuous plating layer, consequently becoming the most commercially used technique for plating from the mid-nineteenth century until today.24
The metals that are commonly applied by electroplating include gold, silver, nickel, chromium, and zinc. The plating can be deposited as pure metal, as successive layers of different metals, or as alloys. Any surface, not only metal, can be electroplated, including plastic, plaster, and resin, if prepared with a conductive coating. Chemical and/or electrolytic treatments have also been used in electroplating to obtain a wider color palette.
Identification
-
Unless the piece is marked as electroplated, it can be a difficult technique to identify.
-
If concealed areas such as the base of a sculpture are gilded, it is likely that the sculpture was electroplated because the effort required to mask these areas during plating would be greater than the value of the gold saved.
-
Visible indicators of electroplating include loss of the thin plating due to wear, and also blistering of the plating layer due to the substrate being imperfectly cleaned or the electroplating bath not being adequately maintained (fig. 296).
-
If nickel is detected analytically through the gilding, it may be a base layer for electroplating.25
-
Electroplating is very often less than 1 µm thick for modern sculptures, but it may exceed 20 µm on nineteenth-century sculptures.
-
If possible, examine a cross section of the plating and substrate layers at high magnification; the plating layer will be even in texture and thickness.
Risks of misidentification/misinterpretation
-
If the plating is very thick, it may be difficult to distinguish gold plating from solid gold by surface analysis.
-
Confusion with amalgam plating may occasionally occur because of the use in the nineteenth and twentieth centuries of mercury salts to prepare the surface, a process known as quicking,26 which leaves small traces of mercury.
-
The electrogilding of iron is preceded by plating with copper. In such cases the under-plating may be detected.
1.2 What other gilding techniques might be found on a
sculpture?
1.2.1 Mechanical plating
Mechanical attachment of metal foil is one of the earliest methods (fig. 297).27 Foil is generally defined as a thin sheet of metal formed by hammering or rolling, and unlike metal leaf, it is thick enough to support its own weight when held at the edge. A sheet of metal foil can be wrapped around an object and held in place by folding and crimping the joints. Alternatively, it can be fixed by rivets or by the edges of the sheet into grooves that are hammered shut or filled with (fig. 298). Inserting gold foil into grooves is a method mentioned by Pliny for a sculpture of the young Alexander.28 Rubbing thin gold or silver sheet into fine grooves or punch marks is a method used on ironwork and copper alloys.29
Identification
-
Foil gilding tends to blur the detail of a casting (fig. 299) and can be detected by the presence of wrinkles or folded overlaps in the plating layer, or the presence of grooves in the casting (fig. 300).
-
On corroded or worn metal surfaces, X-radiography may be necessary to reveal evidence of keying (roughening of the surface) to hold the gilding or silvering; traces of precious metal in the keying will show up because of its greater opacity to X-rays than the baser metal of the substrate.
1.2.2 Diffusion gilding
Diffusion gilding is the attachment of gold foil by heating and burnishing to promote interdiffusion between the gold and the metal substrate, thus creating a metal-to-metal bond without solder. It is difficult to achieve on copper alloys, as a layer of copper oxides forms easily during the heating process, and is a barrier to the plating’s bonding. The technique is more common when gilding silver, both in antiquity prior to the introduction of fire gilding30 and in modern times. It has rarely been identified on copper-alloy statuary, but it has been reported on Roman period bronzes.31
Identification
-
Diffusion gilding can be identified by metallographic examination of a polished cross section. It cannot be identified by surface observation.
-
In section it is recognizable by the bonding of the gilding layer. Effects induced by heat, in particular fine fingers of gold penetrating the surface of the bronze and grains crossing at the interface, will be visible.32 Very fine porosity may also be apparent at the interface.
-
No mercury will be present, and the thickness of the bonded gold-foil layer will be 3–10 µm.
Risks of misidentification/misinterpretation
-
Because of the moderate use of heat, diffusion gilding can be similar to fire gilding in appearance, but analysis will show no mercury.
-
It may also be confused with leaf gilding applied without heat, as outlines of the foils may be seen on the surface, but the penetration of gold into the bronze will not be present unless heat was used.
1.2.3 Depletion gilding
Unlike other gilding processes, depletion gilding is a subtractive process rather than an additive one and can therefore only be used on a copper alloy that contains a significant amount of gold. The process is based on the resistance of gold to oxidation: acids and/or heat are used to remove the copper from the surface of the copper alloy, leaving behind an ultra-thin surface layer of almost pure gold. It is a standard goldsmithing technique33 for improving the golden appearance of baser gold alloys and is known from the third millennium BCE onward.34 But it is rare on sculpture, except on small figures, for example from the pre-Columbian cultures of South and Central America.35 Modern goldsmiths use mineral acids such as sulfuric or nitric, and it is thought that prior to the discovery of these strong acids, organic acids such as oxalic were used. The resulting matte surface is burnished or hammered to create a continuous layer of gold.36
Identification
-
This technique is best detected by examining a polished cross section through the surface plating and measuring the gradient of gold composition from the core metal to the surface by, for example, energy-dispersive spectroscopy (EDX) in a scanning electron microscope (SEM).
-
Burial in a corrosive environment can cause loss of baser metals from the surface of a gold alloy, but in that case the surface will be pitted and porous, not burnished to a smooth, continuous layer of gold.
1.2.4 Electrochemical displacement/replacement plating
Electrochemical displacement/replacement plating uses no external electrical current source. Instead it takes advantage of the natural galvanic potential difference between metals, causing atoms of the metal with the higher galvanic potential, for example gold, to attach themselves to a metal object of lower galvanic potential, for example copper. This method can be used to apply gold, silver, or tin onto copper and has been documented in pre-Columbian metalwork from Peru and Ecuador.37 It is a method commonly used now to restore worn plating, often using proprietary pastes applied by rubbing.38
Identification
-
This form of plating is not suited to large sculpture, but can be useful for restoring areas of worn plating on objects of any size.
-
Because the reaction stops when the surface is completely coated, the plating is extremely thin (typically 0.5–2 µm) and fragile.
No mercury is present.
-
It can be distinguished from fusion plating (see I.7§1.2.5 below) since the plating layer is devoid of any metallographic structures such as dendrites, which are typical of plating applied molten.
-
Electrochemical displacement plating is similar to electroplating, but usually results in a thinner layer.
1.2.5 Fusion plating
Fusion plating is a metal coating applied in molten form, which generally limits its use to small objects or components. The metal best suited to this form of plating is tin (with a melting temperature of 232°C; see I.7§1.3.2 below). Gold and silver are more difficult to apply as molten and are relatively rare, although small Iron Age and Roman items are silvered by this method,39 and some small pre-Columbian cast-copper objects exhibit fusion silvering and gilding.40 Fusion plating of both silver and gold requires the precious metals to be alloyed with copper to lower the melting temperature. In pre-Columbian examples these plating layers were found to have also been treated to remove copper from the gilding or silvering alloys—that is, depletion gilding/silvering took place after the fusion plating was applied.41
Identification
-
As it is applied molten, it has the typical microstructure of a cast alloy (see I.2§3.1).
-
Drips or runs may be visible on the surface, and the plating is likely to be of uneven thickness, but with no mercury present.
-
Loss of copper from the surface of the plating, caused by either corrosion or deliberate removal to improve the color, may also be evident.
1.3 What are the features of silvering and tinning?
1.3.1 Silvering
Many of the methods used for gilding have been used to apply silver to sculpture.42 The commonest is electroplating: the silver can be deposited with a satin or bright metal finish. Mercury silvering is much less common than mercury gilding, especially for statuary, but it was sometimes used in conjunction with mercury gilding in Han dynasty China (206 BCE–220 CE) (fig. 292, see I.7§1.1.2 above).43
Examples of mechanical application of silver-foil plating survive on small statues from such periods as the Late Bronze Age and Roman.44 Foil attached with a continuous layer of tin-lead alloy, a technique of silvering (but not gilding) known as close plating, can be used on small sculpture. Silver coatings applied in molten form are also only suited to small figures such as those found in pre-Columbian South America.45 Silvering with leaf, French plating (multiple layers of leaf applied using heat and pressure), and electrochemical plating are techniques not well documented on sculpture, but may have been used for repairs to plating.
Identification
-
Silvering is much less durable than gilding, and where it survives it may not appear silver in color, so first the metal should be identified by elemental analysis, for example XRF.
-
Techniques of application may be distinguished by the same methods as for the different gilding techniques outlined above.
-
Detection of compounds containing antimony, bismuth, selenium, and sulfur indicate the use of electroplating.
-
The base metal for electroplating with silver is frequently a white metal alloy of nickel, copper, and zinc. This alloy is known by various trade names and may be marked, depending for example on place of origin: EPNS (electroplated nickel silver, UK), Alpaca or Alpacca (South America), Alfenide (used by Christofle for some silver-plated pieces). If plated, the sculpture should not carry the marks “925” or “sterling.”
-
Evidence of mechanical plating may be seen in the form of grooves even where plating is lost (see I.7§1.2.1 above).
-
Examination of a polished cross section through the substrate and the plating layer can be helpful.
Risks of misidentification/misinterpretation
-
White metal coatings other than silver have been widely used, the commonest being tin (see I.7§1.3.2 below).
-
Arsenic-rich surfaces are also silvery in color. A bronze bull statuette from Horoztepe, Anatolia, dating to circa 2100 BCE, has a pale coating of arsenic alloy.46 On small castings of arsenic-copper alloy the silvery appearance is the result of segregation of the arsenic compound from the surface of the casting during cooling.47
1.3.2 Tinning
Tinning dates back to at least the eighth century BCE.48 It is applied to copper, bronze, brass, iron, and steel, either by wiping molten tin onto the heated base metal or by hot dipping into molten tin.49 Tinning can coat whole surfaces with silvery-colored plating or, when applied selectively, can be used to make decorative patterns. It is most commonly found on small items.
Identification
-
Tinned surfaces are frequently mistaken as being silvered from their visual appearance.
-
Elemental analysis can confirm this, but it can be difficult to distinguish tinning from the bright silvery surfaces of cast high-tin bronzes without informed microscopic study of the surface and cross section.50
-
Silvery surfaces are sometimes found on cast bronzes caused by a casting phenomenon known as tin sweat or inverse segregation (fig. 163).51 This may be an accident of casting but could be deliberately encouraged to create a silvery surface on bronze.
2 Why investigate gilding and plating? and other FAQs
2.1 What can I learn from the identification of plating on
a sculpture?
Correct identification can further the understanding of the technological and cultural significance of the object and its makers. Knowing the type(s) and technique(s) of plating may also help to determine the date it was applied and the subsequent history of interventions. It also informs decision making in cultural heritage conservation, for example when assessing the need for a special microclimate to protect a sculpture.
2.2 How easily can I identify whether a bronze has been
plated?
Gilding and plating are often clearly visible, but it may be difficult to see when the plating is worn or concealed under corrosion. Furthermore, bronze and brass are typically golden in color, and areas of clean or varnished metal on a corroded statue can appear to be gilded when no gilding is present. Nevertheless, a combination of microscopic examination and elemental analysis can confirm the presence of plating. It should also be remembered that the plating now visible may not be original. Regilding over the lifetime of a sculpture is common (figs. 301, 302), and several techniques may be encountered on a single piece.
2.3 Are sculptures gilded using pure gold or alloyed gold?
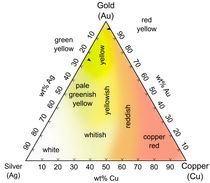
Although gilding can be of pure gold,52 gold alloys with several percent of copper and/or silver have often been used, making the golden color redder or whiter (fig. 303). In addition, imitations are common from at least as early as the medieval period.53 Yellow metal is not necessarily gold, nor is white metal plating necessarily silver, so elemental analysis is advisable.
2.4 What does the presence of mercury prove?
Mercury is used for fire gilding, but it may also indicate the presence of cinnabar, the red mercury sulfide pigment. Quicking from electroplating and some metal polishes may leave traces of mercury, although the residual level of mercury in these is usually too low for XRF to detect.
2.5 Does the absence of mercury prove that mercury gilding
was not carried out?
A large proportion of the mercury will have evaporated during the gilding process but some will remain as mercury compounds, so it is unusual for it to be completely undetectable by XRF analysis.
2.6 Can the thickness and composition of plating layers be
linked to specific workshops?
The thickness and composition of the layers can indicate the plating technique used, but they are unlikely to be helpful in identifying a specific workshop.
2.7 Can traces of lost or hidden metal plating be
detected, and can original and later intervention be
distinguished?
Potentially, yes. Visual examination of the whole surface can reveal evidence, and radiography can detect plating hidden by corrosion, at least when high-density metals such as gold or silver are applied and with sufficient thickness so that they appear whiter than the surrounding bronze on the radiograph. To understand the full history, it may be necessary to take small samples from areas that are believed to represent the sequence of gilding layers for examination in cross section. The layer closest to the cast surface will be the earliest, but if all are applied by the same technique, they may be contemporary to build up thicker plating. The presence of different techniques is a sign of later interventions. Recent leaf gilding may be indicated if cobalt, manganese, or iron compounds are detected by elemental analysis, as in the twentieth century they replaced the lead-based siccative used in early oil-gilding processes. Gold-colored paints and electrochemical plating pastes are frequently used in modern restoration of damaged areas.
2.8 Is white metal plating usually made with silver?
No, it is very often tinning, especially on small sculptures from the Roman period. Arsenic-rich surfaces are also silvery in appearance.54
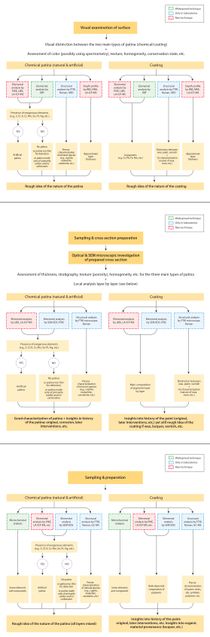
3 Checklist: How do we investigate gilding and plating?
The flowchart in figure 304 will guide you in the visual inspection of plating (see also fig. 305). For more details on how to identify plating processes, refer to the Identification sections in this chapter, which describe all potential processes. For details on analytical techniques see tables 5, 10 Open viewer, and II.6.
XRF surface analysis can determine the composition and thickness of a plating layer, though the best estimate of the thickness is obtained by ion beam analysis (Rutherford backscattering spectrometry). For further investigation, sampling may be required. SEM-EDX analysis of cross sections are often performed, but with high-purity gold the thickness can be overestimated due to smearing caused by mechanical polishing of the soft gold. Ionic polishing helps to avoid this problem. For more, see II.6§1.2.
Keep in mind that plating is often renewed and restored during the lifetime of a sculpture (fig. 289). Traces of lost or hidden metal plating are potentially detectable, and original and later interventions are sometimes distinguishable (see I.7§2.7).
Notes
-
Oddy, W. Andrew. 1993. “Gilding of Metals in the Old World.” In Metal Plating and Patination: Cultural, Technical and Historical Developments, edited by Susan La Niece and Paul Craddock, 171–81. Oxford, London, Boston [etc.]: Butterworth-Heinemann.. For example, only 12 kg of gold was necessary to regild the Dome of Les Invalides, Paris. ↩︎
-
Nicholson, E. D. 1979. “The Ancient Craft of Gold Beating.” Gold Bulletin 12 (4) (December): 161–66.. ↩︎
-
For a brief introduction to contemporary practices in leaf gilding see: https://www.philamuseum.org/learn/conservation/case-study-gilding-diana. ↩︎
-
Darque-Ceretti, Évelyne, Eric Felder, and Marc Aucouturier. 2011. “Foil and Leaf Gilding on Cultural Artifacts: Forming and Adhesion.” Matéria (Rio de Janeiro) 16 (1): 540–59. https://doi.org/10.1590/S1517-70762011000100002.. ↩︎
-
Strahan, Donna K., and Christopher A. Maines. 2000. “Lacquer as an Adhesive for Gilding on Copper Alloy Sculpture in Southeast Asia.” In Gilded Metals: History, Technology and Conservation, edited by Terry Draymann-Weisser, 185–202. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.. ↩︎
-
Darque-Ceretti, Évelyne, Eric Felder, and Marc Aucouturier. 2011. “Foil and Leaf Gilding on Cultural Artifacts: Forming and Adhesion.” Matéria (Rio de Janeiro) 16 (1): 540–59. https://doi.org/10.1590/S1517-70762011000100002.. ↩︎
-
Strahan, Donna K., and Christopher A. Maines. 2000. “Lacquer as an Adhesive for Gilding on Copper Alloy Sculpture in Southeast Asia.” In Gilded Metals: History, Technology and Conservation, edited by Terry Draymann-Weisser, 185–202. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.. ↩︎
-
There is evidence from analysis that animal glue was used for the very fugitive gilded detail on Donatello’s (Italian, ca. 1386–1466) Chellini roundel (ca. 1450) in the V&A (A.1-1976) (Museum departmental records, cited in Motture, Peta. 2019. The Culture of Bronze: Making and Meaning in Italian Renaissance Sculpture. London: V&A Publishing., 68). See also Oddy, W. Andrew, Philippa Pearce, and Lorna Green. 1988. “An Unusual Gilding Technique on Some Egyptian Bronzes.” In Conservation of Ancient Egyptian Materials, edited by Sarah C. Watkins and Carol E. Brown, 35–39. London: UK Institute for Conservation, Archaeology Section.. ↩︎
-
All compositions in this publication are in weight %. ↩︎
-
Theophilus. (ca. 1122) 1979. On Divers Arts: The Foremost Medieval Treatise on Painting, Glassmaking, and Metalwork. Translated by John G. Hawthorne and Cyril Stanley Smith. New York: Dover.; Anheuser, Kilian. 1997. “The Practice and Characterization of Historic Fire Gilding Techniques.” JOM: Journal of the Minerals, Metals & Materials Society 49 (11): 58–62.; Anheuser, Kilian. 1999. Im Feuer vergoldet: Geschichte und Technik der Feuervergoldung und der Amalgamversilberung. AdR-Schriftenreihe zur Restaurierung und Grabungstechnik 4. Stuttgart, Germany: Theiss.. ↩︎
-
Arcet, Jean-Pierre-Joseph d’. 1818. Mémoire sur l’art de dorer le bronze au moyen de l’amalgame d’or et de mercure. Paris: De l’imprimerie de Mme. Veuve Agasse.; Cellini, Benvenuto. 1568 (1967). The Treatises of Benvenuto Cellini on Goldsmithing and Sculpture. Translated by Charles Robert Ashbee. New York: Dover Publications.. ↩︎
-
Lins, Andrew, and W. A. Oddy. 1975. “The Origins of Mercury Gilding.” Journal of Archaeological Science 2:365–73.. ↩︎
-
Cellini, Benvenuto. 1568 (1967). The Treatises of Benvenuto Cellini on Goldsmithing and Sculpture. Translated by Charles Robert Ashbee. New York: Dover Publications., 175; Motture, Peta. 2019. The Culture of Bronze: Making and Meaning in Italian Renaissance Sculpture. London: V&A Publishing., 68. ↩︎
-
Goodison, Nicholas. 1974. Ormolu, the Work of Matthew Boulton. London: Phaidon.. ↩︎
-
Oddy, W. Andrew, Mavis Bimson, and Susan La Niece. 1981. “Gilding Himalayan Images: History Tradition and Modern Techniques.” In Aspects of Tibetan Metallurgy, edited by W. A. Oddy and W. Zwalf, 87–101. British Museum Occasional Paper 15. London: British Museum.; Furger, Alex R. 2017. The Gilded Buddha: The Traditional Art of the Newar Metal Casters in Nepal. Basel, Switzerland, and Frankfurt, Germany: Librum.. ↩︎
-
Oddy, W. Andrew, Mavis Bimson, and Susan La Niece. 1981. “Gilding Himalayan Images: History Tradition and Modern Techniques.” In Aspects of Tibetan Metallurgy, edited by W. A. Oddy and W. Zwalf, 87–101. British Museum Occasional Paper 15. London: British Museum.; Furger, Alex R. 2017. The Gilded Buddha: The Traditional Art of the Newar Metal Casters in Nepal. Basel, Switzerland, and Frankfurt, Germany: Librum.. Cellini did not recommend this method as too much mercury could dull the gold (Motture, Peta. 2019. The Culture of Bronze: Making and Meaning in Italian Renaissance Sculpture. London: V&A Publishing., 68n152). ↩︎
-
Margreiter, Raphael, Jonas Baumann, Ioanna Mantouvalou, Martin Radtke, Uwe Reinholz, and Erik Strub. 2022. “Investigations on Fire-Gilding.” Archaeometry 64 (6): 1465–78. https://doi.org/10.1111/arcm.12797. ↩︎
-
Anheuser, Kilian. 1997. “The Practice and Characterization of Historic Fire Gilding Techniques.” JOM: Journal of the Minerals, Metals & Materials Society 49 (11): 58–62.. ↩︎
-
Because lead does not dissolve in copper and copper alloys but forms separate inclusions, it risks weeping out when the surface is heated for the amalgam process (lead melts at a low temperature). See Oddy, W. Andrew, Mavis Bimson, and Susan La Niece. 1981. “Gilding Himalayan Images: History Tradition and Modern Techniques.” In Aspects of Tibetan Metallurgy, edited by W. A. Oddy and W. Zwalf, 87–101. British Museum Occasional Paper 15. London: British Museum.; Oddy, W. Andrew. 2000. “A History of Gilding with Particular Reference to Statuary.” In Gilded Metals: History, Technology and Conservation, edited by Terry Draymann-Weisser, 1–19. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.; Vincent, Brice, David Bourgarit, and Paul Jett. 2012. “Khmer Bronze Metallurgy during the Angkorian Period (12th–13th Centuries): Technical Investigation of a New Selected Corpus of Artifacts from the National Museum of Cambodia, Phnom Penh.” In Scientific Research on Ancient Asian Metallurgy, edited by Paul Jett, Blythe McCarthy, and Janet G. Douglas, 124–53. London: Archetype.. ↩︎
-
Arcet, Jean-Pierre-Joseph d’. 1818. Mémoire sur l’art de dorer le bronze au moyen de l’amalgame d’or et de mercure. Paris: De l’imprimerie de Mme. Veuve Agasse.; Jett, Paul. 1993. “A Study of the Gilding of Chinese Buddhist Bronzes.” In Metal Plating and Patination: Cultural, Technical and Historical Developments, edited by Susan La Niece and Paul Craddock, 193–200. Oxford, London, Boston [etc.]: Butterworth-Heinemann.; Strahan, Donna K., and Christopher A. Maines. 2000. “Lacquer as an Adhesive for Gilding on Copper Alloy Sculpture in Southeast Asia.” In Gilded Metals: History, Technology and Conservation, edited by Terry Draymann-Weisser, 185–202. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.; Cowell, Michael, Susan La Niece, and Jessica Rawson. 2003. “A Study of Later Chinese Metalwork.” In Scientific Research in the Field of Asian Art: Proceedings of the First Forbes Symposium at the Freer Gallery of Art, edited by Paul Jett, 80–89. London: Archetype.. ↩︎
-
Oddy, W. Andrew, Mavis Bimson, and Susan La Niece. 1981. “Gilding Himalayan Images: History Tradition and Modern Techniques.” In Aspects of Tibetan Metallurgy, edited by W. A. Oddy and W. Zwalf, 87–101. British Museum Occasional Paper 15. London: British Museum.. ↩︎
-
La Niece, Susan. 1990. “Silver Plating on Copper, Bronze and Brass.” Antiquaries Journal 70 (1): 102–14.. ↩︎
-
Hunt, L. B. 1973. “The Early History of Gold Plating.” Gold Bulletin 6 (1): 16–27. https://doi.org/10.1007/BF03215178.; Raub, Christoph. 1993. “The History of Electroplating.” In Metal Plating and Patination: Cultural, Technical and Historical Developments, edited by Susan La Niece and Paul Craddock, 284–290. Oxford, London, Boston [etc.]: Butterworth-Heinemann.; Lins, Andrew. 2000. “Gilding Techniques of the Renaissance and After.” In Gilded Metals: History, Technology and Conservation, edited by Terry Draymann-Weisser, 241–66. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.. ↩︎
-
Oddy, W. Andrew. 1995. “A History of Gilding with Particular Reference to Statuary.” In Proceedings of the Objects Specialty Group Session 23rd Annual Meeting in St. Paul, Minnesota, June 10, 1995. Objects Specialty Group Postprints. http://resources.conservation-us.org/osg-postprints/postprints/v03/oddy/.; Grant, Alistair, and Angus Patterson. 2018. The Museum and the Factory: The V&A, Elkington and the Electrical Revolution. V&A 19th-Century Series. London: Lund Humphries.. ↩︎
-
Selwyn, A. 1950. The Retail Jeweller’s Handbook. London: Heywood.. ↩︎
-
Lins, Andrew, and Sally Malenka. 2000. “The Use of Mercury Salts in Gold Electroplating.” In Gilded Metals: History, Technology and Conservation, edited by Terry Draymann-Weisser, 267–82. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.. ↩︎
-
Oddy, W. Andrew. 2000. “A History of Gilding with Particular Reference to Statuary.” In Gilded Metals: History, Technology and Conservation, edited by Terry Draymann-Weisser, 1–19. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.. ↩︎
-
Pliny the Elder. 1857. Natural History, translated by John Bostock and Henry Thomas Riley. London: H.G. Bohn. https://www.google.com/books/edition/The_Natural_History_of_Pliny/9zwZAAAAYAAJ?hl=en&gbpv=0., 34.19n94. ↩︎
-
Oddy, W. Andrew. 1993. “Gilding of Metals in the Old World.” In Metal Plating and Patination: Cultural, Technical and Historical Developments, edited by Susan La Niece and Paul Craddock, 171–81. Oxford, London, Boston [etc.]: Butterworth-Heinemann.. The Queen Napirasu (1340–1300 BCE) in the Louvre (inv. Sb 2731) also shows large grooves that probably have been used for gilding. Examples are known also for Khmer bronzes (Vincent, Brice. 2012. “Samrit. Étude de la métallurgie du bronze dans le Cambodge angkorien (fin du XIe–début du XIIIe siècle).” PhD diss., Université Paris 3. http://www.theses.fr/2012PA030032., 288–90). ↩︎
-
Oddy, W. Andrew, Susan La Niece, and Nigel D. Meeks. 1981. “Diffusion-Bonding as a Method of Gilding in Antiquity.” MASCA Journal 1 (8): 239–41.. ↩︎
-
See Bott, Kati, and Frank Willer. 2014. “Gold – Ein unvergängliches Material: Vergoldungstechniken an Bronzestatuen.” In Gebrochener Glanz: römische Großbronzen am UNESCO-Welterbe Limes, edited by Stephanie Müller, 207–9. Mainz, Germany: Nünnerich-Asmus.; Willer, Frank, Roland Schwab, and Manuela Mirschenz. 2017. “Ergebnisse der archäometrischen und herstellungstechnischen Forschungen and den ‘Limesbronzen.’” In Römische Grossbronzen am UNESCO-Welterbe Limes, edited by M. Kemkes, 79–105. Darmstadt, Germany: Konrad. https://landesmuseum-bonn.lvr.de/media/lmb/forschung/bilder_4/restaurierung/Mirschenz_M.Schwab_R._Willer_F._Ergebnisse_der_archaeometrischen_und_herstellungstechnischen_Forschungen_an_den_Limesbronzen.pdf.. ↩︎
-
Bott, Kati, and Frank Willer. 2014. “Gold – Ein unvergängliches Material: Vergoldungstechniken an Bronzestatuen.” In Gebrochener Glanz: römische Großbronzen am UNESCO-Welterbe Limes, edited by Stephanie Müller, 207–9. Mainz, Germany: Nünnerich-Asmus.; Willer, Frank, Roland Schwab, and Manuela Mirschenz. 2016b. Römische Bronzestatuen am Limes: Archäometrische Untersuchungen Zur Herstellungstechnik.” Bonner Jahrbücher 216:57–187. https://journals.ub.uni-heidelberg.de/index.php/bjb/article/view/70701/64059.; Willer, Frank, Roland Schwab, and Manuela Mirschenz. 2017. “Ergebnisse der archäometrischen und herstellungstechnischen Forschungen and den ‘Limesbronzen.’” In Römische Grossbronzen am UNESCO-Welterbe Limes, edited by M. Kemkes, 79–105. Darmstadt, Germany: Konrad. https://landesmuseum-bonn.lvr.de/media/lmb/forschung/bilder_4/restaurierung/Mirschenz_M.Schwab_R._Willer_F._Ergebnisse_der_archaeometrischen_und_herstellungstechnischen_Forschungen_an_den_Limesbronzen.pdf.. ↩︎
-
Grimwade, Mark. 1999. “The Surface Enrichment of Carat Gold Alloys: Depletion Gilding.” Gold Technology 26:16–23.. ↩︎
-
La Niece, Susan. 1995. “Depletion Gilding from 3rd Millennium BC Ur.” Iraq 57:41–47.. ↩︎
-
Lechtman, Heather. 1984. “Pre-Columbian Surface Metallurgy.” Scientific American 250 (6): 56–63.; Scott, David A. 1983. “Depletion Gilding and Surface Treatment of Gold Alloys from the Narino Area of Ancient Colombia.” Journal of the Historical Metallurgy Society 17 (2): 99–115.; Scott, David A. 2000. “A Review of Gilding Techniques in Ancient South America.” In Gilded Metals: History, Technology and Conservation, edited by Terry Drayman-Weisser, 203–22. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.. ↩︎
-
Grimwade, Mark. 1999. “The Surface Enrichment of Carat Gold Alloys: Depletion Gilding.” Gold Technology 26:16–23.. ↩︎
-
Lechtman, Heather. 1979. “A Pre-Columbian Technique for Electrochemical Replacement Plating of Gold and Silver on Copper Objects.” JOM: Journal of the Minerals, Metals & Materials Society 31 (12): 154–60.; Scott, David A. 2000. “A Review of Gilding Techniques in Ancient South America.” In Gilded Metals: History, Technology and Conservation, edited by Terry Drayman-Weisser, 203–22. London: Archetype in association with the American Institute of Conservation of Historic and Artistic Works.. ↩︎
-
Stalker, John, and George Parker. 1960. A Treatise of Japanning and Varnishing. Chicago: Quadrangle Books.. ↩︎
-
Northover, J. Peter, and Chris J. Salter. 1990. “Decorative Metallurgy of the Celts.” Materials Characterization 25:109–23.; La Niece, Susan. 1993. “Silvering.” In Metal Plating and Patination: Cultural, Technical and Historical Developments, edited by Susan La Niece and Paul Craddock, 201–10. Oxford, London, Boston [etc.]: Butterworth-Heinemann.. ↩︎
-
Scott, David A. 1986. “Fusion Gilding and Foil Gilding in Pre-Hispanic Colombia and Ecuador.” In Metalurgia de America precolombina, edited by Clemencia Plazas de Nieto, 283–325. Bogotá, Colombia: Banco de la Republica.. ↩︎
-
Scott, David A. 1983. “Depletion Gilding and Surface Treatment of Gold Alloys from the Narino Area of Ancient Colombia.” Journal of the Historical Metallurgy Society 17 (2): 99–115.. ↩︎
-
La Niece, Susan. 1990. “Silver Plating on Copper, Bronze and Brass.” Antiquaries Journal 70 (1): 102–14.. ↩︎
-
La Niece, Susan. 1993. “Silvering.” In Metal Plating and Patination: Cultural, Technical and Historical Developments, edited by Susan La Niece and Paul Craddock, 201–10. Oxford, London, Boston [etc.]: Butterworth-Heinemann.. ↩︎
-
La Niece, Susan. 1990. “Silver Plating on Copper, Bronze and Brass.” Antiquaries Journal 70 (1): 102–14.. ↩︎
-
Scott, David A. 1986. “Fusion Gilding and Foil Gilding in Pre-Hispanic Colombia and Ecuador.” In Metalurgia de America precolombina, edited by Clemencia Plazas de Nieto, 283–325. Bogotá, Colombia: Banco de la Republica.. ↩︎
-
Museum of Fine Arts Boston, 58.14; Smith, Cyril Stanley. 1970. “An Examination of the Arsenic-Rich Coating on a Bronze Bull from Horoztepe.” In Application of Science in Examination of Works of Art, edited by William J. Young, 96–102. Boston: Museum of Fine Arts, Boston.. ↩︎
-
Craddock, Paul T. 1981. “The Copper Alloys of Tibet and Their Background.” In Aspects of Tibetan Metallurgy, edited by W. Andrew Oddy and Wladimir Zwalf, 1–33. British Museum Occasional Paper 15. London: British Museum.; La Niece, Susan. 1989. “White Copper.” Journal of the Historical Metallurgy Society 23 (1): 9–15.. ↩︎
-
Meeks, Nigel. 1993. “Surface Characterization of Tinned Bronze, High-Tin Bronze, Tinned Iron and Arsenical Bronze.” In Metal Plating and Patination: Cultural, Technical and Historical Developments, edited by Susan La Niece and Paul Craddock, 247–75. Oxford, London, Boston [etc.]: Butterworth-Heinemann.. ↩︎
-
Welter, Jean-Marie. 2019. “Contact Tinning: A Millennia-Old Plating Technology.” Archaeometry 61 (10): 906–920.. ↩︎
-
Meeks, N. D. 1986. “Tin-Rich Surfaces on Bronze: Some Experimental and Archaeological Considerations.” Archaeometry 28 (2): 133–62.. ↩︎
-
Meeks, N. D. 1986. “Tin-Rich Surfaces on Bronze: Some Experimental and Archaeological Considerations.” Archaeometry 28 (2): 133–62.. ↩︎
-
Here pure gold means unalloyed gold. ↩︎
-
Smith, Cyril Stanley, and John G. Hawthorne. 1974. Mappae Clavicula: A Little Key to the World of Medieval Techniques. Philadelphia: American Philosophical Society.. ↩︎
-
Smith, Cyril Stanley. 1970. “An Examination of the Arsenic-Rich Coating on a Bronze Bull from Horoztepe.” In Application of Science in Examination of Works of Art, edited by William J. Young, 96–102. Boston: Museum of Fine Arts, Boston.; Craddock, Paul T. 1981. “The Copper Alloys of Tibet and Their Background.” In Aspects of Tibetan Metallurgy, edited by W. Andrew Oddy and Wladimir Zwalf, 1–33. British Museum Occasional Paper 15. London: British Museum.. ↩︎