38. New Approaches in Stabilizing Chloride-Contaminated Ancient Bronzes Using Corrosion Inhibitors and/or Electrochemical Methods to Preserve Information in the Patinas
- Vasilike Argyropoulos, Technological Educational Institute of Athens, Department of Conservation of Antiquities and Works of Art
- Soussani Mavroforaki, Technological Educational Institute of Athens, Department of Conservation of Antiquities and Works of Art
- Maria Giannoulaki, Technological Educational Institute of Athens, Department of Conservation of Antiquities and Works of Art
- Stamatis C. Boyatzis, Technological Educational Institute of Athens, Department of Conservation of Antiquities and Works of Art
- Thanasis Karabotsos, Technological Educational Institute of Athens, Department of Conservation of Antiquities and Works of Art
- Aggeliki Zacharopoulou, Department of Materials Science and Engineering, School of Chemical Engineering, National Technical University of Athens
- Elodie Guilminot, EPCC Arc’Antique, Nantes, France
Abstract
Introduction
Ancient bronzes often contain surface decoration in the corrosion layers or show evidence of tinning, niello, gilding, paint, or organic materials in the corrosion layers. Depending on the quantity of chlorides in their surfaces, traditional conservation methods to stabilize bronze disease are either used locally to treat the artifact with Ag2O particles or zinc dust, with complete immersion in sodium sesquicarbonate (pH 10) solutions with or without electrolysis to convert cupreous chloride to more stable corrosion product(s);1 and/or incorporate the use of corrosion inhibitors, such as benzotriazole.2 Our goal was to develop alternative conservation methods to treat such objects in order to preserve information by minimizing the overall changes that may occur to the patina surfaces with such treatments.
The paper presents a novel treatment approach for a thirteenth-century Byzantine copper frying pan that was excavated from a shipwreck off of Rhodes and retains what appears to be intentional tinning on its surface. The paper examines the black surface to determine its nature and the condition of the artifact. It further discusses the treatment of the object with the aim of preserving this black layer, which serves as an archaeological record providing evidence of its use in food preparation. Traditional chloride removal methods for marine copper-alloy artifacts may result in unwanted removal of these types of compounds/corrosion products. Our treatment involved the application of local electrolysis with 1% (w/v) of sodium sesquicarbonate solution on areas with no evidence of tinning, and the application of L-cysteine as a nontoxic corrosion inhibitor to help preserve the artifact over the long term.
Local Electrolysis and L-Cysteine as a Corrosion Inhibitor for Ancient Copper Alloys
The Metals Conservation Lab (TEI-A) in Egaleo, Greece, carried out research from 2006 to 2015 in collaboration with the National Technical University of Athens (NTUA) and Arc’Antique of Nantes, France, to develop the methods of local electrolysis of metals to either extract chlorides or reduce corrosion products back to their metal state during treatments. This approach offers better control for the conservator than the traditional methods that use complete immersion of marine artifacts in electrolytes involving either chemical or electrochemical methods. In 2008, TEI-A was commissioned to treat an eight-ton iron paddle wheel recovered from the 1868 shipwreck Patris in the Aegean Sea. To extract chlorides, conservators used a cost-effective method that has been traditionally used by industry to stabilize reinforced concrete. The scientific results both in the laboratory and in the field on the paddle wheel (in a dry state) indicated that the technique is capable of removing the majority of the chlorides with two applications of impressed current (applied as local electrolysis) for 24 hours each time.3 The method was also applied to remove chlorides locally from a wet marine copper-alloy composite artifact with textile remaining on the surface, applying 1% (w/v) sodium sesquicarbonate solutions to selected areas.4 Using this localized approach, areas on the artifacts with organic remains could be avoided and thus preserved.
L-cysteine has already been investigated in metals conservation as a replacement for benzotriazole (BTA) applied to corroded bronze coupons (with nantokite formation) by immersion in water at 0.15M for 24 hours;5 by immersion in ethanol at 0.01M for 24 hours;6 and during PEG400 treatments for a marine copper-alloy composite artifact with organic remains.7 Gravgaarda and van Lanschot (2012) also carried out tests on real bronze artifacts in comparing cysteine to BTA. All these studies found color changes to the corrosion layers (graying effect) with the cysteine application, which darkens to a black color with longer immersion exposures. With their electrochemical measurements, Abu-Baker et al. (2013) found that, with increasing amounts of tin in the bronze alloy, the strength of the inhibitor’s chemisorption on the surface of the alloy, or its corrosion inhibition efficiency, increases. Further research found that immersion in cysteine for copper alloys results in producing cystine from the oxidation of cysteine, where the presence of iron or copper ions serves as a catalyst in this reaction. This oxidation reaction results in reducing metal ions, which form a metal ion cystine complex on the surface of the material, and producing either a soluble or insoluble cysteine/cysteinate complex with metal ions and cystine precipitate respectively in the solutions.8 The graying effect is most likely a result of the metal ion cystine complex on the surface.
Scientific Investigation of a Marine Byzantine Frying Pan
The frying pan was exposed to an open-sea environment as well as buried in the marine sediment for centuries. As a result, there has been substantial loss of metal with cracks and deformation to its shape. The pan was made from a copper sheet of approximately 98 wt.% copper and 1 wt.% tin (analyzed by scanning electron microscopy with energy dispersive X-ray analysis [SEM-EDAX]; see below), which was hammered into shape with a base diameter of around 19 to 21 cm as shown in figure 38.1. A riveted 10.5 cm handle also made of copper sheet metal was folded and hammered into a tube shape. Given the poor condition of the object, it is difficult to assess whether the sides of the pan were straight or angled; they have a preserved maximum height of 3.7 cm. The interior of the object is covered with a black surface (see fig. 38.1), which is assumed to be evidence of intentional tinning. Copper cooking wares were always intentionally tinned, usually with a hot-tinning process in their interior, to prevent the copper from contaminating food during cooking.

Corrosion samples were analyzed non-destructively from five different locations with a two-step process: first, SEM-EDAX to identify the micromorphology of the compounds/corrosion products on the surface as well as their elemental concentrations; second, X-ray diffraction (XRD) analysis to identify their mineralogical composition. SEM-EDAX type GEAN GSM 6510 low vacuum with X-ART detector (working distance: 20 mm) was used. Also, XRD analysis was carried out using the Olympus BTXII Benchtop XRD/XRF (Co source) with software (XPowder) for processing the resulting XRD data (XRD range 5–55o 2Ø), including the AMSCD mineral database. The sample for XRD analysis needs to be around 15 mg and running times from 20 to 60 minutes depending on the type of sample. Also, a very small piece from the base of the pan containing metal and corrosion, which was separated from the main object due to burial conditions, was examined using a stereomicroscope with digital camera.
Figures 38.2 and 38.3 show the microscopic images of this cross-section of the copper metal with the black surface as well as other types of corrosion products. In both images, there appears to be a thin tin metal or intermetallic compound covering the copper metal. Manti and Watkinson (2011) concluded that such black surfaces are the result of intentional tinning rather than surface corrosion of low-tin archaeological bronzes.

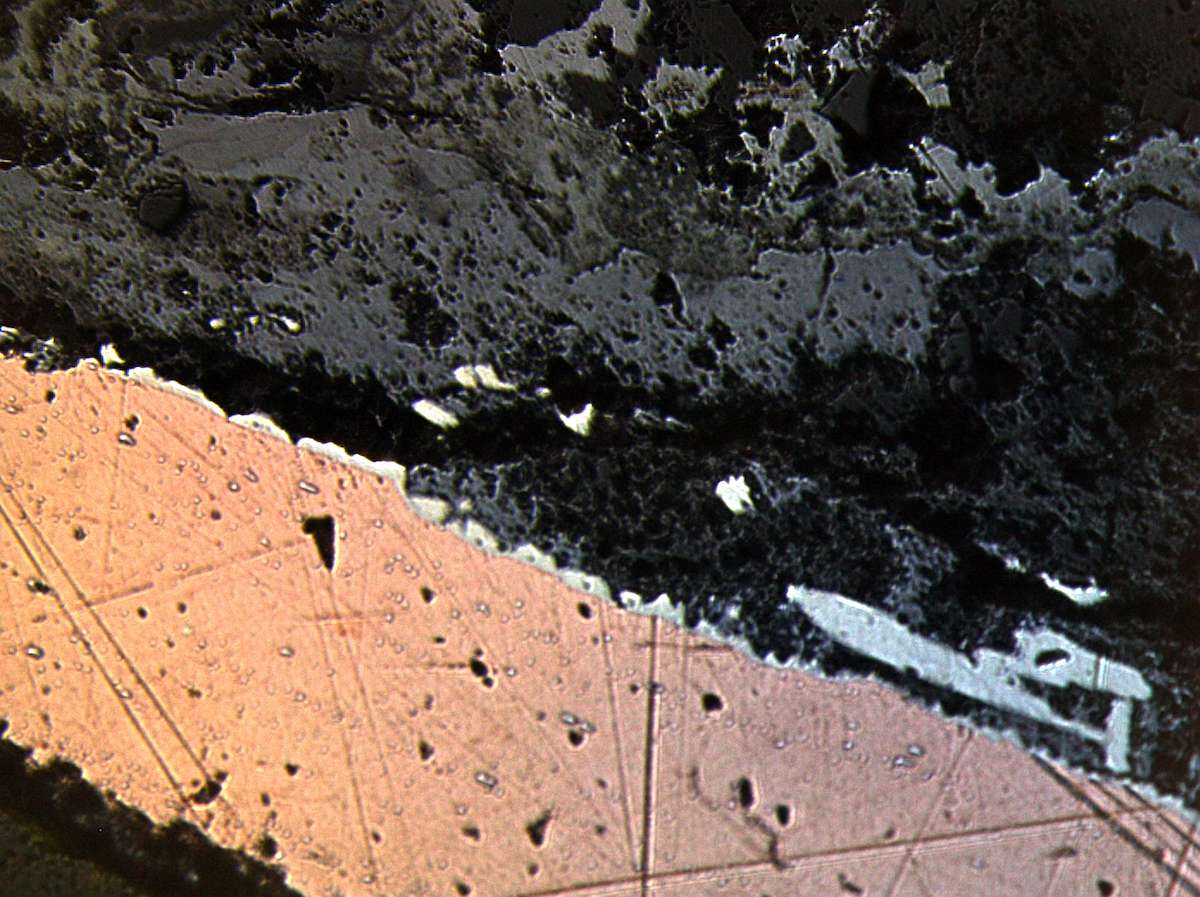
Black corrosion products from another area of the interior surface were further analyzed using SEM-EDAX and XRD. Figure 38.4 shows the micromorphology of the corrosion products, with small round globules indicating the possibility of intermetallic compound η-Cu6Sn5 with around a 57 wt.% tin concentration, suggesting a hot-tinning process. However, XRD was unable to confirm this compound due to the small sample size and the complexity of the diffraction pattern due to the fact that it was difficult to isolate this compound from other mineral species present in the sample.

XRD analyses of corrosion taken from five different areas on the object found copper oxides, chlorides, carbonates, and sulphides: tenorite, cuprite, clinoatacamite, paratacamite, malachite, chalcocite, covellite, and digenite. The presence of copper sulfides indicates that the object was buried under marine sand, with the eventual result that the main cathodic reaction was hydrogen evolution stimulated by sulfate-reducing bacteria.9 An adherent protective film of copper sulfides resulted on the surface. The large holes in the surface of the frying pan indicate that pitting corrosion was prevalent due to the presence of cuprous chlorides (nantokite), although its presence was not confirmed by XRD analysis. Nantokite is found next to the metal, and sampling may not have reached the area containing this corrosion product, since it is always present in wet marine copper alloys.
Treatment Approach
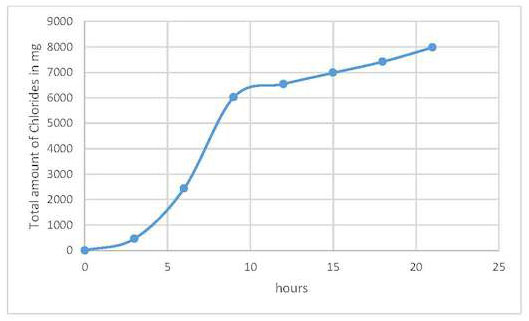
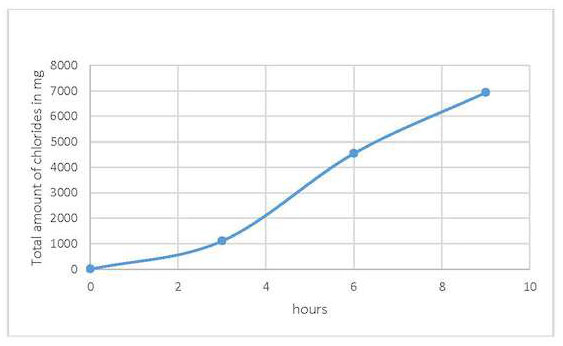
Cathodic polarization was carried out at a constant potential of –0.76 V vs. SSE using an EG&G Verstat II potentiostat (applied as local electrolysis) for 3 hours each time before changing the electrolyte (1% w/v sodium sesquicarbonate solutions) in the sponge to remove chlorides locally from the frying pan on selected areas (fig. 38.5). The areas treated were the base of the pan and its handle, which did not show any evidence of tinning (black surface). In this way, areas on the artifact with tinned surface could be avoided by the procedure and thus preserved. The artifact was maintained wet during treatment with deionized water. Cathodic polarization was applied to the handle a total of seven times (for 3 hours each time) until a minimum quantity of chlorides was found in the electrolyte solution, each time changing the sponge and using fresh electrolyte solution. However, for the base of the pan the procedure was repeated 3 times for 3 hours each time. The number of applications also depended on the visual changes to the patina after each application of local electrolysis. After the end of the treatment procedure, the object was thoroughly rinsed with deionized water.

After each application, the electrolytic solution used was collected to check the amount of chlorides extracted during the electrolysis, using the HANNA chloride electrode and Mohr’s method for chloride determination. The quantities of chlorides removed after each 3-hour application of cathodic polarization were plotted vs. time and are shown in figures 38.6 and 38.7 for the handle and base, respectively. The results show that the technique removed around 7 and 8 g of chlorides from each surface, respectively. The electrolytic treatment could have continued, since the slope of each plot continued to increase, but it was decided to stop the treatment and use L-cysteine instead so as to minimize surface patina changes. After the electrolysis, any calcium carbonate concretions from the object’s surface were removed using dental tools, and finally cysteine as a corrosion inhibitor was applied to ensure the object would be stable after treatment.
Cysteine was tested on another fragment of a tinned copper-alloy artifact from the same marine site. The piece was immersed in 1% (w/v) solution of cysteine in deionized water for 3 hours as opposed to 24 hours as tested by the other studies. After slight rinsing with a soft brush, it was placed in a climatic chamber for 24 hours at 70% RH with an untreated fragment from the same artifact, for comparative purposes. The cysteine-treated piece was found to be stable while the other had signs of active corrosion. For the frying pan, it was decided to apply the cysteine with a paintbrush every 2 hours over 3 days. Again after light rinsing with a soft brush, it was left to dry in a polystyrene box on top of polyethylene foam, with silica gel to help dry it gradually. Figure 38.8 shows the base of the frying pan before (a) and after (b) treatment, and indicates that, apart from a slight graying effect, both electrolysis and the application of L-cysteine maintained the patina appearance. One year after treatment, the frying pan remains stable in storage with no signs of active corrosion. The conservation work for the object still needs to be completed by deciding whether a coating will be used as well as the best method for restoring it for future display in a museum in Rhodes.
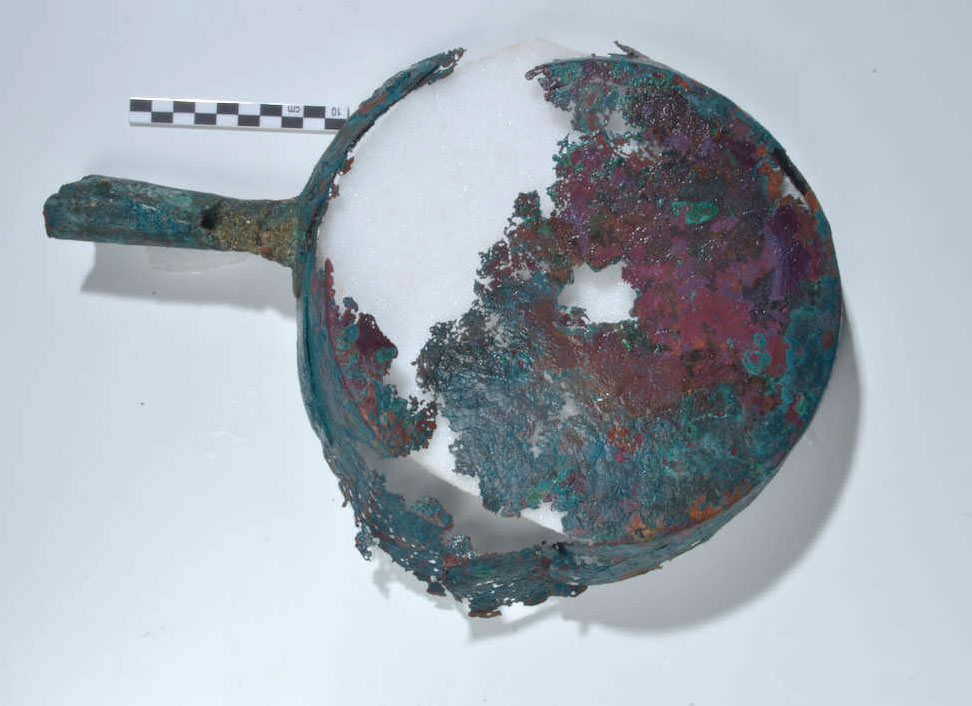

Conclusions
The paper presented a case study in which local electrolysis and the use of L-cysteine, an alternative corrosion inhibitor to BTA, can successfully be applied to stabilize a marine copper-alloy artifact with evidence of a tinned surface. Such a treatment approach can be applied to marine copper-alloy artifacts when the aim is to preserve information in the patina of such objects, which may traditionally be sacrificed during conservation treatment in order to stabilize the artifact. The treatment approach was successful at stabilizing a thirteenth-century marine copper-alloy frying pan with evidence of tinning.
Acknowledgments
The authors would like to thank P. Manti for her useful comments regarding the analyses of the tinned surface. We also thank G. Koutsoflakis from the Ephorate of Underwater Antiquities for his support of our work. The authors would also like to acknowledge that this conservation work was supported by the MERMAID research project that was operated under the THALES action and coordinated by the TEI-A. The research on cysteine and local electrolysis has been co-financed by the European Union (European Social Fund – ESF) and Greek national funds through the Operational Program “Education and Lifelong Learning” of the National Strategic Reference Framework (NSRF) - Research Funding Program: ARCHIMEDES III: Investing in the knowledge society through the European Social Fund.
Notes
- Adriaens and Dowsett 2013. ↩
- Cano and Lafuente 2013. ↩
- Argyropoulos and Batis 2013. ↩
- Argyropoulos et al. forthcoming. ↩
- Gravgaarda and van Lanschot 2012. ↩
- Abu-Baker et al. 2013 ↩
- Zacharopoulou et al. 2016; Argyropoulos et al. forthcoming. ↩
- Argyropoulos et al. forthcoming. ↩
- North and MacLeod 1987, 82. ↩
Bibliography
- Abu-Baker et al. 2013
- Abu-Baker, A. N., I. D. MacLeod, R. I. Sloggett, and R. Taylor. 2013. “A Comparative Study of Salicyaldoxime, Cysteine, and Benzotriazole as Inhibitors for the Active Chloride-based Corrosion of Copper and Bronze Artifacts.” European Scientific Journal 9 (33): 229–51.
- Adriaens and Dowsett 2013
- Adriaens, A., and M. Dowsett. 2013. “Electrochemical Analysis of Metallic Heritage Artefacts: Time-lapse Spectroelectrochemical Techniques.” In Corrosion and Conservation of Cultural Heritage Metallic Artefacts. Cambridge: Woodhead.
- Argyropoulos and Batis 2013
- Argyropoulos, V., and G. Batis. 2013. “Saving a Marine Iron Paddle Wheel Removed from the 1868 Steam Engine Shipwreck ‘Patris’ during an Economic Crisis in Greece.” Big Stuff Conference. Ottawa, 25–27 Sep, 2013. http://cstmuseum.techno-science.ca/doc/content/cstm/argyropouloubigstuffpaper.pdf
- Argyropoulos et al. forthcoming
- Argyropoulos, V., S. Boyatzis, M. Giannoulaki, A. Malea, A. Pournou, S. Rapti, A. Zacharopoulou, and E. Guilminot. Forthcoming. “Preliminary Investigation of L-cysteine as a Corrosion Inhibitor for Marine Composite Artefacts Containing Copper or Iron Alloys.” ICOM-CC WOAM 2016. Florence.
- Cano and Lafuente 2013
- Cano, E., and D. Lafuente. 2013. “Corrosion Inhibitors for the Preservation of Metallic Heritage Artefacts.” In Corrosion and Conservation of Cultural Heritage Metallic Artefacts, ed. P. Dillmann, D. Watkinson, E. Angelini, and A. Adriaens, 570–90. Cambridge: Woodhead.
- Gravgaarda and van Lanschot 2012
- Gravgaarda, M., and J. van Lanschot. 2012. “Cysteine as a Non-toxic Corrosion Inhibitor for Copper Alloys in Conservation.” Journal of the Institute of Conservation 35(1): 14–24.
- Manti and Watkinson 2011
- Manti, P., and D. Watkinson. 2011. “Hot-tinning of Low Tin Bronzes.” In METAL 2010: Proceedings of the Interim Meeting of the ICOM-CC Metal Working Group, ed. P. Mardikian, C. Chemello, C. Watters, and P. Hull, 92–98. Clemson: ICOM-CC and Clemson University.
- North and MacLeod 1987
- North, N. A., and I. D. MacLeod. 1987. “Corrosion of Metals.” In Conservation of Marine Archaeological Objects, ed. C. Pearson, 68–98. Oxford: Buttersworth.
- Zacharopoulou et al. 2016
- Zacharopoulou, A., G. Batis, V. Argyropoulos, and E. Guilminot. 2016. “The Testing of Natural Corrosion Inhibitors Cysteine and Mature Tobacco for Treating Marine Composite Objects in PEG400 Solutions.” Journal of Conservation Science 7 (1): 259–64.